
The ‘platypus’ enzyme to help develop new, more effective antibiotics
14. 08. 2023
The development of new antibiotics is one of the global health challenges of our time. Now, in an international collaboration with the University of Tokyo, researchers from the Institute of Microbiology of the CAS have decoded the structure of a key condensation enzyme used for antibiotic biosynthesis and have clarified how it works at the molecular level. The enzyme is one-of-a-kind, according to the researchers, because it combines the individual components in a unique way. The scientists liken the enzyme to the duck-billed platypus, which also appears as if it were a cross of various animals. The study, “Molecular basis for carrier protein-dependent amide bond formation in the biosynthesis of lincosamide antibiotics”, was published in Nature Catalysis.
The substances, which are also effective against the dreaded Staphylococcus aureus and other dangerous bacteria, have been in development for several years by researchers from the Czech Academy of Sciences in collaboration with the Czech company Santiago Chemicals. They are based on research that began in 2017 under the leadership of Jiří Janata, head of the Laboratory for Biology of Secondary Metabolism at the Institute of Microbiology of the CAS.
At that time, the researchers came up with a new, more effective antibiotic called CELIN, which combines the building blocks of two natural substances – celesticetin and lincomycin. Combining lincomycin with the less active celesticetin had never been considered before. Chemically, it is a very complicated process and for decades scientists lacked the necessary knowledge. Today, collaborations have resulted in dozens of successful derivatives, several of which are also effective against antibiotic-resistant bacteria.
The Staphylococcus aureus can cause many different diseases, such as skin infections, the cold, fever, and sore throat. It can also cause abdominal pain, diarrhoea, and vomiting.
Now, as part of a project at the National Institute of Virology and Bacteriology, experts have made another breakthrough by elucidating the structure of a unique enzyme in cooperation with the team of Professor Ikura Abe from the University of Tokyo. This enzyme is essential for the preparation of celesticetin, a bacteriostatic antibiotic.
The enzyme, which will help in the advancement of new antibiotics effective against resistant bacteria, combines individual components in a unique way. Its structure is unlike any known protein. At a molecular level, it can be compared to the platypus, which also appears as if it were a cross of various animals.
“Between 2015 and 2018, we made significant contributions to deciphering the remarkable biosynthesis of the lincosamide antibiotics celesticetin and lincomycin, in which the individual building blocks assemble like a molecular version of a Lego set. When we tested these principles during the construction of the CELIN hybrid, it occurred to us that our building capabilities would be significantly more extensive if we understood the function of the essential enzymes in the biosynthesis of natural lincosamides,” Jiří Janata explains.
Exceptional results thanks to Czech–Japanese cooperation
Already from the sequence of the enzymes under study (the order of the amino acids), it became evident that they would differ to a great degree from all previously known proteins. “I was fascinated by the key condensation enzyme. It was unlike anything else we’d seen before. Intuition in biology tells us that it is unique systems that bring the most profound surprises and breakthrough discoveries. I approached Professor Abe, who focuses on unusual enzymes of the so-called special or secondary metabolism of natural substances, and we agreed to work together on deciphering the structure and function of the condensation enzyme we’ve just published about,” Janata explains, describing the origin of the collaboration with the Japanese group. This has gradually expanded to include three other proteins that are key to the folding and processing of the building blocks of antibiotics.
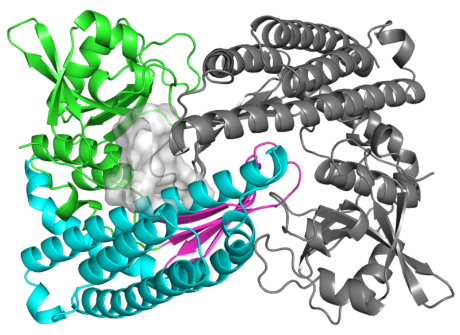
The structure of the enzyme is unlike any known protein. Researchers liken it to the platypus, which also appears as if it were a cross of various animals.
A difficult and precarious path to success
The structure of the basic form of the condensation enzyme was decoded by Stanislav Kadlčík from the Institute of Microbiology of the CAS, who was a postdoctoral fellow in the Tokyo research group in 2019. “Before the structure of a protein can be studied, the protein must be purified and crystallised, which is a long and unpredictable task with no certainty of success. Each protein requires specific conditions. If you are lucky and manage to obtain a protein crystal of a quality that is sufficient for measuring the structure, all is not won yet and you are a mere one third of the way there. To decipher the mechanism of function of an unusual protein you must also have crystals with enzyme substrates, often just as unusual and therefore not readily available. And then, modified forms of the enzyme must be prepared and tested. Such research can take several years, and you can stumble upon an obstacle at any time,” Kadlčík says of the demanding work.
The way forward is learning the language of microorganisms
Comparing the assembling of an antibiotic from the individual building blocks to a Lego set is no coincidence. Antibiotics are special, tailor-made molecular constructions that fit precisely into the fundamental biological structures of microorganisms. That’s why they are so effective only against bacteria and are harmless to humans. Antibiotic resistance somehow breaks this tight-fitting jigsaw puzzle and the antibiotics stop working.
Antibiotic resistance expert Gabriela Balíková Novotná from the Laboratory for Biology of Secondary Metabolism adds, “In nature, antibiotics and antibiotic resistance have existed side by side for millions of years – and yet, antibiotics have endured. That’s because antibiotics and resistance are two integral parts of a broader system of chemical communication of microorganisms or, in simplified terms, bacteria.”
In her view, if we can decode these principles from nature, if we can, so to speak, learn the language of bacteria, it will allow us to treat infectious diseases in a completely new way in the future. For instance, it will not be necessary to kill infectious agents with antibiotics. Instead, it will be possible to send signals to the pathogenic bacteria that will persuade them to retreat. Rather than the extermination war aspect antibiotics use has today, new treatment methods may look more like a type of information warfare.
*
The study in question, “Molecular basis for carrier protein-dependent amide bond formation in the biosynthesis of lincosamide antibiotics”, was published in Nature Catalysis.
Prepared by: Markéta Wernerová, Division of External Relations, CAO of the CAS, drawing on the press release of the CAS
Translated by: Tereza Novická, Division of External Relations, CAO of the CAS
Photo: Shutterstock (2); Institute of Microbiology of the CAS
 The text is released for use under the Creative Commons license.
The text is released for use under the Creative Commons license.
Read also
- A trapped state: The pandemic impact on public attitudes, trust, and behavior
- Aerial archaeology: Tracing the footsteps of our ancestors from the sky
- Archaeologists uncover ancient finds along Prague Ring Road
- Our microbiome largely depends on what we eat, says microbiologist Michal Kraus
- The ABCs of writing: Why did its invention mark a turning point for humankind?
- We learn, remember, forget… What can memory actually do? And can we outsmart it?
- New Center for Electron Microscopy in Brno opens its doors to global science
- The hidden lives of waste: What can we learn from waste workers and pickers?
- A unique lab is hidden right beneath Prague’s Vítkov Hill
- Renewables are a strategic investment in European security, scientists say
The Czech Academy of Sciences (the CAS)
The mission of the CAS
The primary mission of the CAS is to conduct research in a broad spectrum of natural, technical and social sciences as well as humanities. This research aims to advance progress of scientific knowledge at the international level, considering, however, the specific needs of the Czech society and the national culture.
President of the CAS
Prof. Eva Zažímalová has started her second term of office in May 2021. She is a respected scientist, and a Professor of Plant Anatomy and Physiology.
She is also a part of GCSA of the EU.